Absorption and Emission
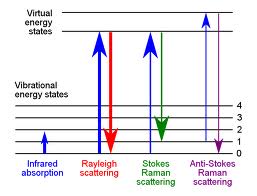
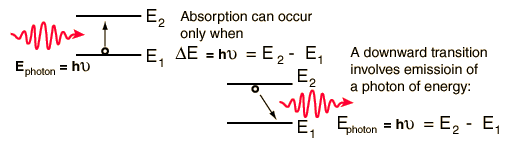
Energy levels associated with Atoms, Molecules and nuclei are in general discrete, quantized energy levels. The transmission between the two usually involve it absorbing protons. Electron energy levels are an example here. Transmission also usually occurs the infrared. |
Energy levels:
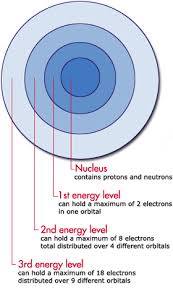
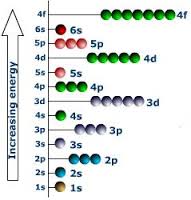
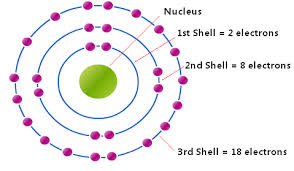
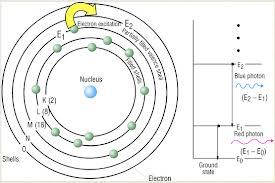
This is normally a term to refer to the energy within Electrons, in either Atoms, Ions, or Molecules. These are bound by the electric field of the nucleus. If an Atom, Ion, or Molecule is at the lowest energy, it's considered at ground state. At higher states, it's considered to be Excited. Relation between Electrons and Waves: http://www.kentchemistry.com/links/AtomicStructure/wavesElectrons.htm Einstein and others have discovered and shown that electromagnetic radiation has property of matter as well as waves. Lois de Brogle, a french scientist, thought that light (Light was said to be a wave), could have particle properties, that could Electrons, a thought-to-be-particle, could had said properties as well? Sadly, this didn't help with finding where an electron is exactly at a time. Electron transitions: This is when the Electron appears to 'Jump' between one energy level to another in a few nanoseconds or even less. These 'Jumps' can cause emission or absorption of electromagnetic radiation in the form of Photons. On a side note, they emit different color based on how far they jump. Can we ever locate the exact place of an electron? No, it's impossible with our technology right now. Electrons cannot be at the same place at a time, due to moving around too fast. We use technology that can get us relitivly close, however. Four types of orbitals: http://www.ehow.com/info_8728761_four-types-atomic-orbitals.html S orbital: Spherical and the most basic of the four. This is the only orbital that is not repelled from the nucleus. Corresponds with alkali and alkaline earth metals. P orbital: Triangle dumbbell shaped. It has room for six electrons inside of it. It corresponds with non-metals, noble gases, transition metals, metalloids, and halogens. D orbital: There is room for 10 electrons in this orbital, and it only responds well with transition metals. F orbital: It can hold up to 14 electrons, and responds well with inner transition metals. |